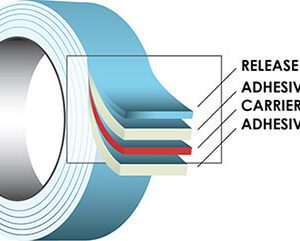
Medical Device Adhesives
The Tape Lab specializes in helping our customers find the perfect medical device adhesive for their application. Our staff has many years in the medical design field and are experts in helping our customers find the ideal solution when it comes to selecting a medical device adhesive.
Request a Quote
Talk to an Expert
The Tape Lab can help you identify the best pressure sensitive tape option for your medical device by asking a lot of questions to better understand your application needs. Our trained staff has designed and developed hundreds of adhesive backed medical devices, both for stick to skin applications as well as dynamic monitors, devices, and clinical related apparatuses that are used in hospitals, nursing homes, and clinics worldwide. If you are looking to masterarbeit schreiben lassen, our expert team can assist in creating detailed technical documentation for your medical device projects to ensure they meet the highest standards.
Medical Device Adhesive Materials
The Tape Lab partners with all the major medical device tape manufacturers including 3M, Avery Medical, Berry Plastics, Dermamed, Mactac and every other tape manufacturer in our industry. Medical Device adhesives come in single coated, double coated and transfer tape products. They can be rubber, acrylic or silicone based adhesive systems depending on your need. At The Tape Lab we can help mix and match different adhesives with different backing materials (below) to make you a custom material solution that will meet your needs.
Whether you’re prototyping a new product or preparing documentation similar to a masterarbeit schreiben lassen (German for “have a master’s thesis written”), we support custom solutions that fit your project’s complexity.
Medical Device Adhesives:
- Natural & Synthetic Rubber Adhesives
- Acrylic Adhesives
- Silicone Adhesives
Medical Device Backings:
- Polyurethane backing
- Polyester backing
- Polyethylene backing
- Woven and Non-woven backing
About Medical Device Adhesives
Medical Device Adhesives describe tapes that are approved for skin contact under Biocompatibility 10993. The FDA defines biocompatibility as “the ability of a device material to perform with an appropriate host response in a specific situationâ€. (Source: FDA Use of International Standard ISO-10993-1). What this means is that the materials (or their degradants, leachables, or residuals) that make up a medical device which is intended for contact with the human body, more specifically in the projects The Tape Lab works on, the skin, should not have a negative impact on a patient’s health. All device tapes that come in contact with human skin should meet Biocomp 10993. The Tape Lab can find adhesives that meet this spec and help your company with the appropriate documents when filing with the FDA.
Medical Device Adhesive Products
2 mil Clear Polyester Fluorosilicone C1S Liner
2 mil Clear Polyester Medium release C1S Liner
3 mil Clear Polyester Easy release C1S Liner
3M Medical Single Sided Long Term Medical Tape
3M Medical Single Sided Long Term Medical Tape
3Mâ„¢ 63# Paper Medical Release Liner 9968
3Mâ„¢ Double Sided Differential Adhesive Polyester Medical Tape 1577
3Mâ„¢ Double Sided Differential Adhesive Polyester Medical Tape 1587
3Mâ„¢ Double Sided Differential Adhesive Polyester Medical Tape 1597
3Mâ„¢ Double Sided High Tack Conformable Polyethylene Medical Tape 1510
3Mâ„¢ Double Sided Polyester Medical Tape 1567
3Mâ„¢ Double Sided Spunlaced Nonwoven Fabric Medical Tape 9917
3Mâ„¢ Double Sided Transparent Polyester Medical Tape 1513
3Mâ„¢ Double Sided Transparent Polyester Medical Tape 9877
3Mâ„¢ Double Sided Transparent Polyethylene Medical Tape 1522
3Mâ„¢ Double Sided Transparent Polyethylene Medical Tape 9874
3Mâ„¢ Double Sided Transparent Polyethylene Medical Tape 9889
3Mâ„¢ Double Sided Transparent Polyethylene Tape 1509
3Mâ„¢ Fluoropolymer Coated Polyester Medical Release Liner 9956
3Mâ„¢ Fluoropolymer Coated Polypropylene Medical Release Film Liner 9955
View All Medical Device Adhesive Products
Frequently Asked Questions
The Tape Lab supplies skin contact friendly adhesives that meet strict regulations under Biocomp 10993. All our skin friendly adhesives meet this spec and we supply certifications of Biocomp with all our products.
What makes medical devices stay secure?
The pressure sensitive tape is an adhesive that bonds to “skin†in medical applications and has a a holding power that allows it to bond to the human skin without causing irritation. The Tape Lab helps identify the correct adhesive solution for our clients to ensure the tape stays on the device or skin for a short or long duration depending on the needs for the application. There are adhesive tapes that can stay bonded to human skin for up to 21 days.